mAbs and Recombinant Proteins
CUSTOMER CHALLENGE
Monoclonal antibodies (mAbs) and other recombinant proteins biopharmaceuticals require specialized QC testing and characterization within a regulated GMP environment. Finding the right partner to accelerate your product’s journey to the patient, ensuring safety and efficacy, can be challenging. This is particularly true when seeking alternatives to traditional culture or animal-based testing. Our new lab excels in mAb characterization, CHO cell line characterization, mAb and recombinant testing, making us the ideal partner for your biotesting needs.
PATHOQUEST SOLUTION
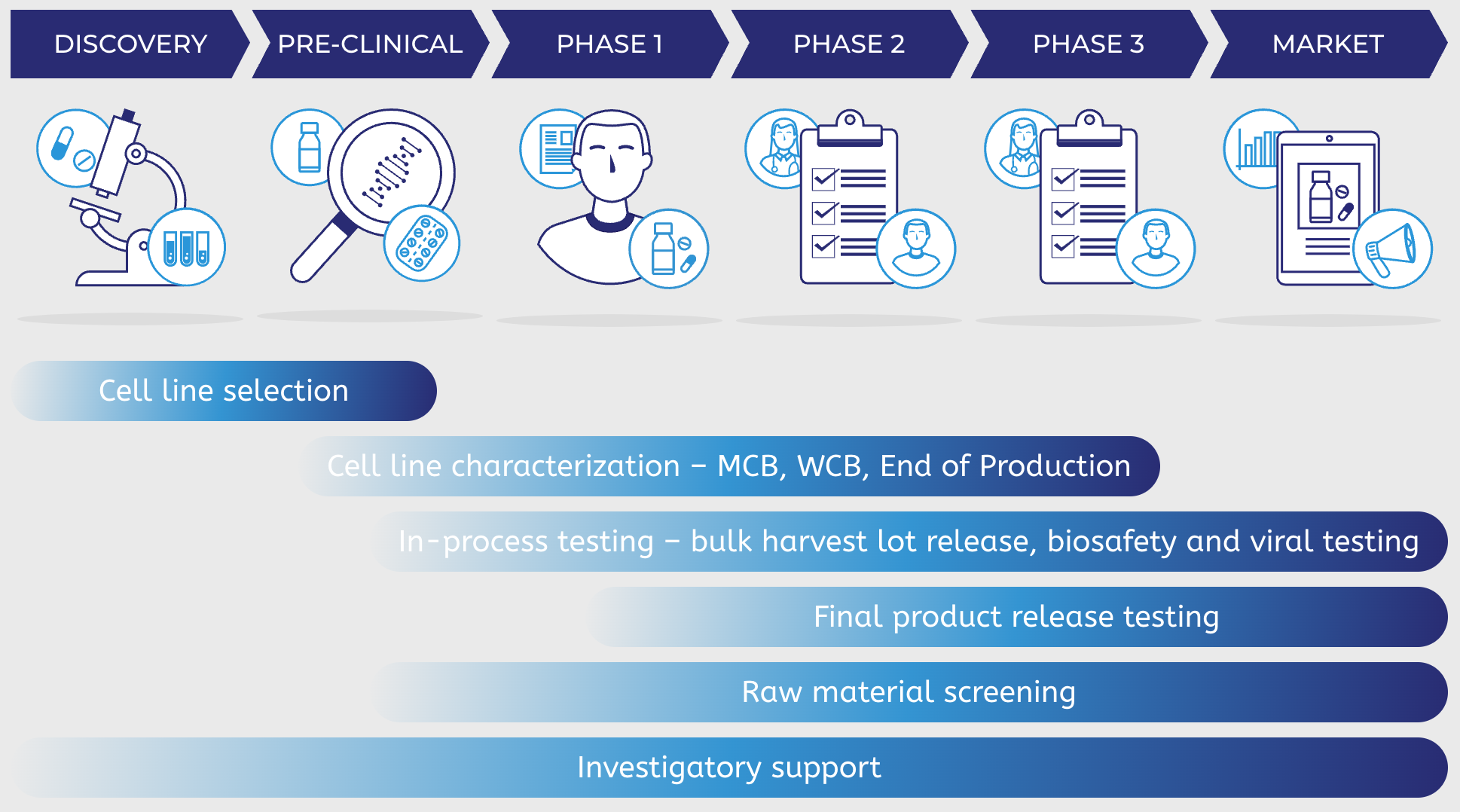
PathoQuest can help characterize, QC and release your recombinant mAb across the manufacturing journey
DISCOVERY
PRE-CLINICAL
PHASE 1
PHASE 2
PHASE 3
MARKET
Cell line selection
Cell line characterization – MCB, WCB, End of Production
In-process testing – bulk harvest lot release, biosafety and viral testing
Final product release testing
Raw material screening
Investigatory support
OUR EXPERTISE
PathoQuest remains at the forefront of NGS for biologic characterization and biosafety testing, especially in the ethical replacement of animal models. Our expertise extends to mAb characterization, CHO cell line characterization, mAb and recombinant testing, We actively participate in regulatory and expert groups, associations, and consortia, as well as frequently publishing in peer-reviewed journals. We are leaders both in innovation and acceptance of NGS technology, while continuously improving the characterization and biosafety testing of biopharmaceuticals.
CHALLENGES SOLVED
PathoQuest can give you more confidence in manufacturing and release of your biopharmaceutical product by addressing key concerns:
- Faster cell line development and clone selection with deeper genetic insights
- De-risking your manufacturing process by rapidly screening critical raw materials such as serum
- Reducing the cell line characterization bottleneck, allowing you to get therapy into clinic faster
- Rapidly identify and resolve manufacturing events such as adventitious agent contamination
- Meeting corporate 3Rs ethical objectives by removing animal models
Modalities
mAbs and Recombinant Proteins
Bacterial and mammalian produced proteins, hormones and peptides
READ MORE
Viral Vectors
Viral gene delivery, oncolytic and immunotherapy including manufacturing plasmids
READ MORE
Cell Therapies
Including gene modified, or unmodified stem cell therapies, allogeneic or autologous
READ MORE
Vaccines
Inactivated, live-attenuated, recombinant, RNA and viral vector products
READ MORE
RNA
Immunotherapies, antiviral, vaccines, RNAi and CRISPR based gene editing
READ MORE
Cultivated Meat
Engineered cells and tissues cultured in more ethical in vitro environments
READ MORE
Contact us
U.S.
466 Devon Park Dr
Wayne, PA 19087
United States
Sign up for our latest news
Follow Us
France
+33 (0)1 70 82 17 90
Biopark -Bâtiment B,
11, rue Watt
75013 Paris, France
Sign up for our latest news
Follow Us
How can PathoQuest help?