In Vivo Replacement
NGS as an ethical alternative to animals in biosafety testing and characterization.


What is in vivo replacement and why is it important?
The use of animal models in the characterization and biosafety testing of biologic medicines has been a staple for decades. Despite their limitations, in vivo testing has remained essential in biologic cell line and viral vector characterization due to the need for prior knowledge of contaminants when using PCR or ELISA. Agnostic cell culture-based methods also fall short as they detect a narrow range of adventitious pathogens. Consequently, animal models, despite their limited sensitivity in detecting a broad range of viruses, have continued as a crucial complementary method.
However, the landscape is changing with the advent of alternatives to animal testing, such as in vitro testing . These in vitro assay methods align with the 3Rs of animal research (Replacement, Reduction, and Refinement), aiming to minimize animal use in scientific research. In vitro testing methods are increasingly recognized for their potential to provide reliable and sensitive detection of contaminants without the ethical and practical issues of animal models.
Next-generation sequencing (NGS) has been used for agnostic detection of unknown pathogens in clinical settings and commercial biopharmaceutical manufacturing, especially for contamination investigations. Moreover the ICHQ5A (R2) and European Pharmacopoeia encourages the replacement of in vivo testing with NGS. “
In collaboration with Charles River Laboratories, PathoQuest has become the first GMP Contract Research Organization (CRO) to demonstrate conclusively that iDTECT® NGS assays are superior to conventional in vivo biosafety testing and characterization methods. This breakthrough, along with regulatory bodies’ commitment to eliminate animal models in most submissions, signals that now is the ideal time to transition from in vivo testing and embrace NGS testing methods as the new standard for biosafety and characterization in biopharmaceuticals.
What we do
Modalities Tested:
- mAbs and recombinant proteins biologics
- Viral vectors
- Cell therapies
- Vaccines
- RNA based therapeutics
- Cultured meat
Benefits of iDTECT® NGS assays
✓ GMP validated – in vivo testing mostly GLP✓ Fast results – days vs weeks✓ Much more reliable and robust than animal models✓ No need to generate neutralizing antibodies✓ Specific identification of contamination✓ Meet corporate 3Rs objectives
Our Assays:
- iDTECT® Virome
- iDTECT® Transcriptome
WHY THIS APPROACH
ICH: Draft Guidline for Consultation Q5A (R2) viral safety evaluation of biotechnology products derived from cell lines of human or animal origin
Publication: Historical evaluation of the in vivo adventitious virus test and its potential for replacement with next generation sequencing (NGS)
Publication: Systematic evaluation of in vitro and in vivo adventitious virus assays for the detection of viral contamination of cell banks and biological products
PathoQuest in vivo Brochure
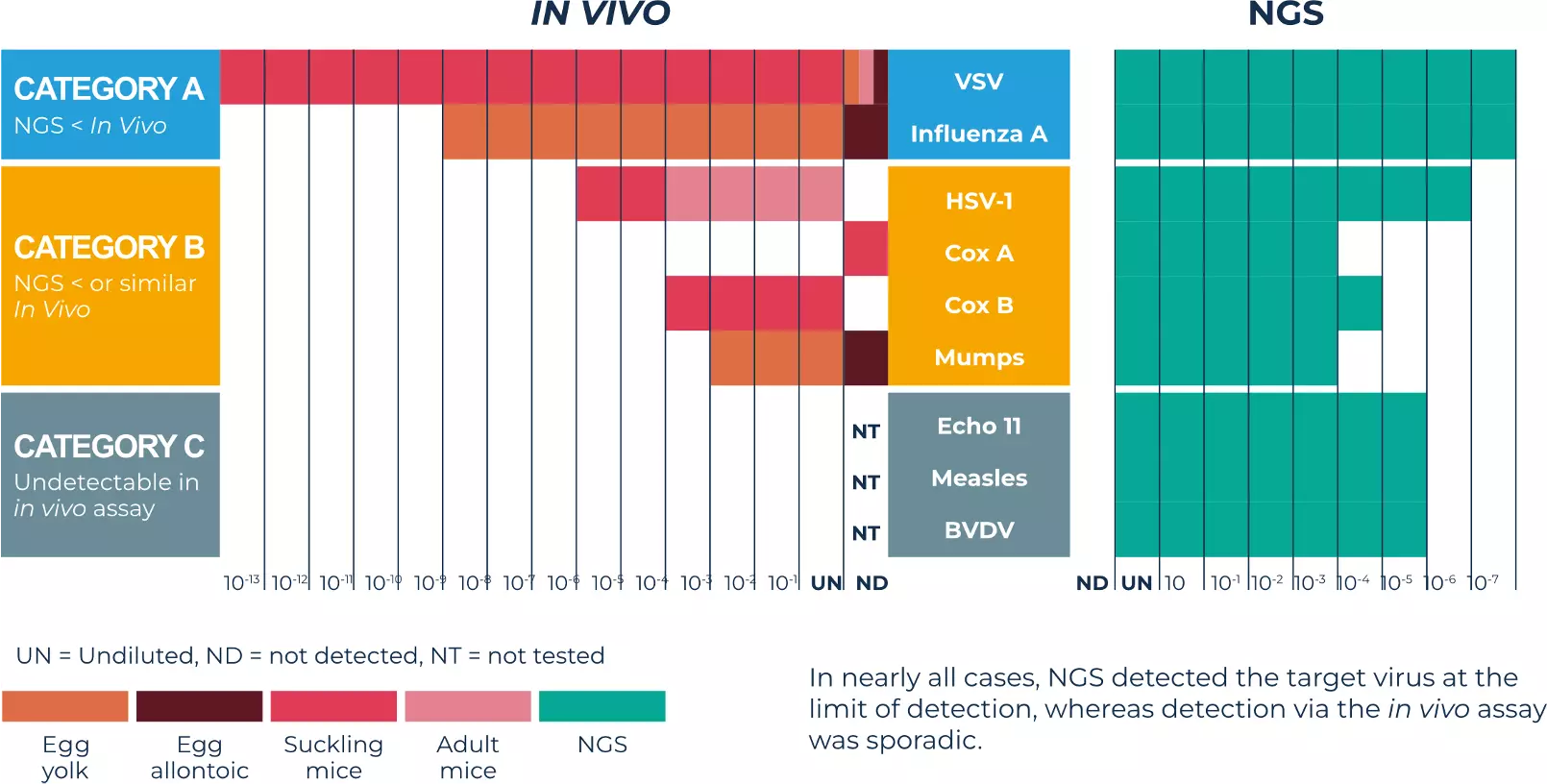
Our comparison data
In vivo assays were performed using suckling mice, adult mice, and embryonated hens’ eggs. For ethical purposes, we chose not to perform all in vivo tests. For each virus category (defined in this publication), only the most sensitive reported ones were used.
Challenges solved
- Difficulty in generating a neutralizing antibody
- Incompatibility of test material with in vivo methods
- Limited sample volume for in vivo characterization
- Meeting corporate objectives to reduce animal use
OTHER SERVICES
Adventitious Virus Testing
Detection of viral contamination within the manufacturing process and beyond.
READ MORE
Integration Site Analysis
Characterisation of genetic modifications for clone selection, genetic stability and lot release
Identity Confirmation
Genetic characterization of viral and plasmid products for release.
READ MORE
Cell Line Characterization
Biosafety screening and stability testing of manufacturing cells.
READ MORE
HLA Genotyping
Characterizing and screening for novel and emerging cell therapies.
READ MORE
Raw Material Testing
Screening of high-risk inputs such as animal products and media.
READ MORE
Contact us
France
+33 (0)1 70 82 17 90
Biopark -Bâtiment B,
11, rue Watt
75013 Paris, France
How can PathoQuest help?
U.S.
466 Devon Park Dr
Wayne, PA 19087
United States
France
+33 (0)1 70 82 17 90
Biopark -Bâtiment B,
11, rue Watt
75013 Paris, France
How can PathoQuest help?
Sign up for our latest news
